CLIA Screen In-Vitro™
5 Panel CSI Cup CLIA Screen In-Vitro™ Rapid Drug Test Cup, No THC, 25/Box | CSI-CUP-955
Free FedEx Ground shipping on orders of $150 or more
Sales Tax Exempt? Call 800-915-7017 before ordering
- SKU:
- CSI-CUP-955
- Availability:
- SHORT DATED LOT - 12-31-2026
Description
5 Panel CSI Cup - CLIA Screen In-Vitro™ Drug Test, CLIA Waived, without THC, 25/Case
SHORT DATED LOT - 12-31-2026
The best-selling CSI Cup (CLIA Screen In-Vitro™) Multi-Panel Drug Test Cup is a No Step, fully integrated, self-contained multi-drug screening cup. No tipping or turning required. Without extra handling steps, the 5 Panel CSI Cup CLIA Screen In-Vitro Rapid Drug Test streamlines urine collection and multi-panel screening in one device. Read results in 5 minutes. The cup is FDA Cleared and CLIA Waived. This cup is a fast visual, competitive panel immunoassay that can be used for the simultaneous, qualitative detection of multiple drug metabolites at specific cutoff levels in human urine. 5 Panel CSI Cup CLIA Screen In-Vitro™ Rapid Drug Test is an all-in-one screening device designed for professional use in workplace and clinical environments.
Features & Benefits
- CLIA Waived

- FDA 510(K) Approved
- OTC - Over-the-Counter Approved for Home Use
- Health Canada Approved

- Sold by the Case of 25 tests
- The Click Seal cap is securely sealed when you hear the “CLICK”
- No step cup
- Up to 99% Accurate
- Dual scale Temperature Strip verifies if the urine is "fresh" and not diluted
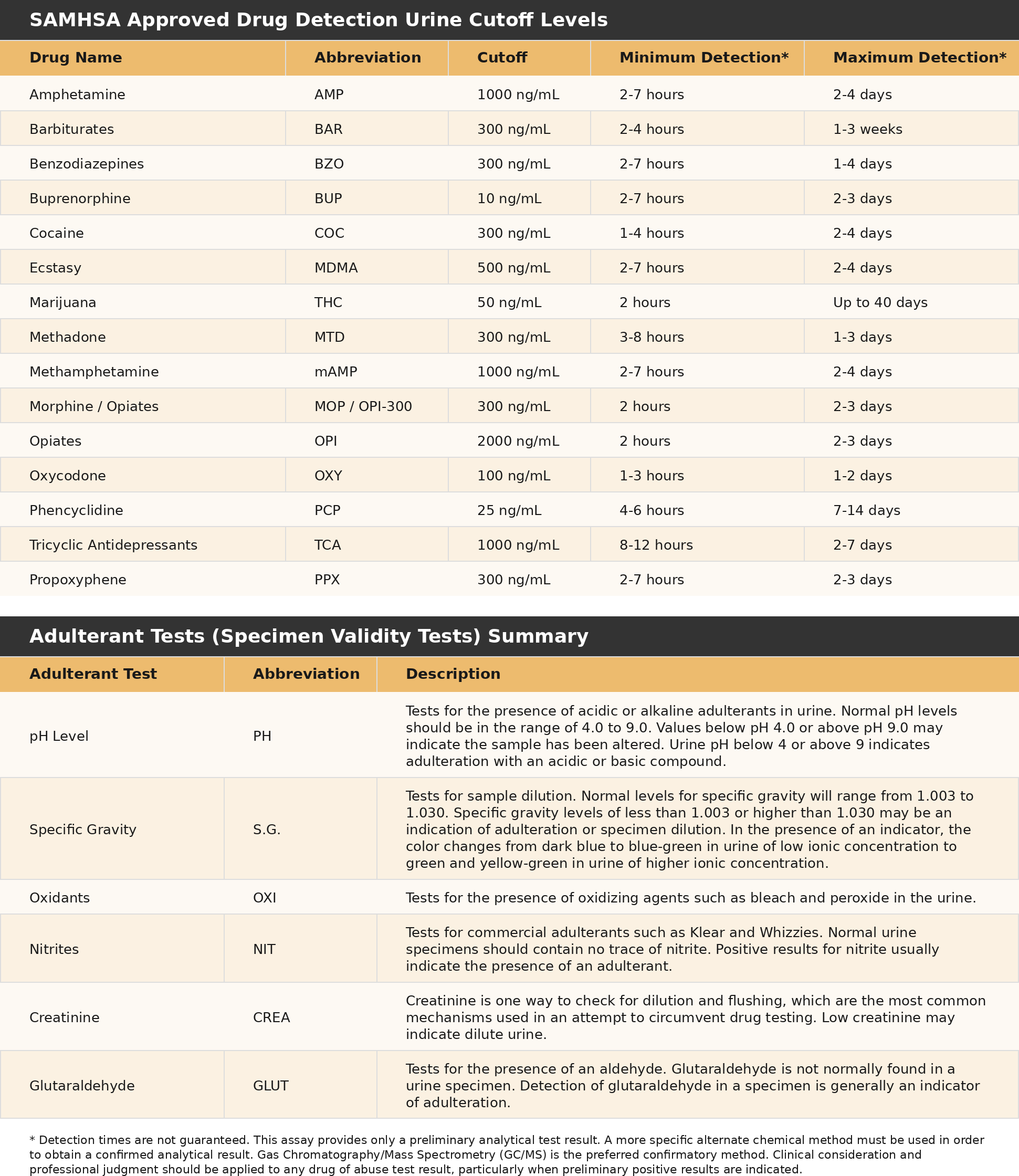
- Adheres to the SAMHSA Cutoff Levels
- Immediate Results
- Shelf-life up to 24 months
- New easy-to-open top for the confirmation lab
Download & Print Reference Materials

![]() CLIA Screen In Vitro Cup (CLIA Waived) Drug Test Package Insert
CLIA Screen In Vitro Cup (CLIA Waived) Drug Test Package Insert

![]() CLIA Screen In Vitro Cup (CLIA Waived) Drug Test Procedure Card
CLIA Screen In Vitro Cup (CLIA Waived) Drug Test Procedure Card

![]() CLIA Screen In Vitro Cup (CLIA Waived) Drug Test Product Flyer
CLIA Screen In Vitro Cup (CLIA Waived) Drug Test Product Flyer
| 5 Panel Configuration | |||
| Item Number | Drugs Tested | Adulterants Tested | Approvals |
| CSI-CUP-955 | BUP10, BZO300, COC300, MOP300, MTD300 (NO THC) | None | FDA 510(k) Cleared CLIA Waived OTC - Over the Counter Health Canada Approved |